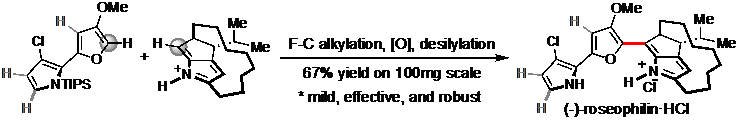梁广鑫课题组介绍
| 课题组长 |
|
| 研究介绍 |
1) 具有重要生物活性或生理功能的复杂天然产物的全合成
2) 药物及精细化学品的合成工艺研究
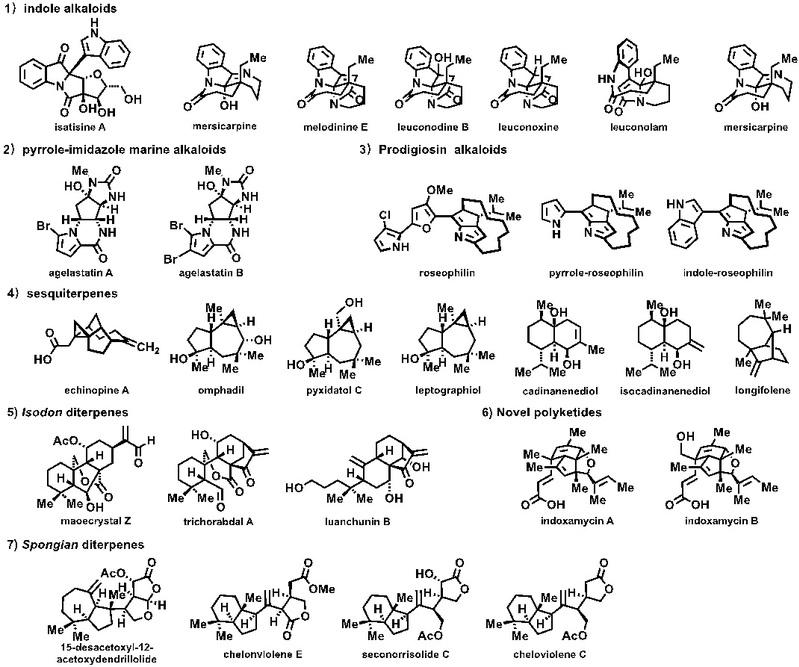
课题组目前完成的天然产物分子:
本课题组长期诚聘博士后研究员,研究助理,硕博连读研究生及联合培养研究生。有意申请者请直接与课题组长联系。
1) Qiao, T.; Wang, Y.; Zheng, S.; Kang, H.; Liang, G.* “Total Syntheses of Norrisolide-Type Spongian Diterpenes Cheloviolene C, Seconorrisolide B, and Seconorrisolide C” Angew. Chem. Int. Ed. 2020, asap. (DOI: 10.1002/anie.202005600)
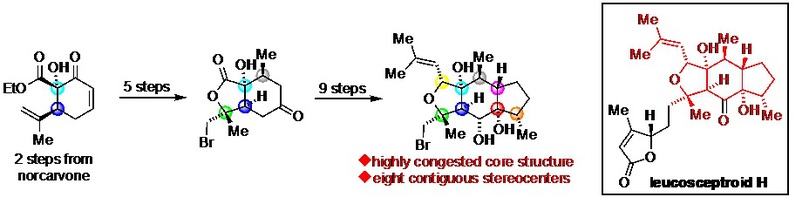
2) Sun, P.; Zhang, C.; Qiao, T.; Ao, J.; Wu, S.; Liang, G.* “Stereoselective Construction of the Highly Congested Tricyclic Core Structure in Leucosceptroid H” Org. Lett. 2020, asap. (DOI: 10.1021/acs.orglett.0c01672)
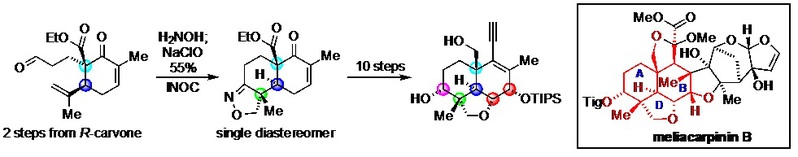
3) Dong, C.; Qiao, T.; Xie, Y.; Zhang, X.; Ao, J.; Liang, G.* “Rapid Construction of the ABD Tricyclic Skeleton in Meliacarpinin B from Carvone Enabled by an INOC Strategy” Org. Chem. Front.2020, asap. (DOI: 10.1039/D0QO00576B)
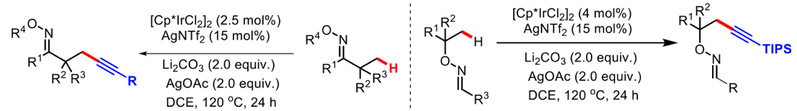
4) Li, X.; Liang, G.*; Shi, Z.* “Regio-Divergent C-H Alkynylation with Janus Directing Strategy via Ir(III) Catalysis”Chin. J. Chem. 2020, 38, 929.
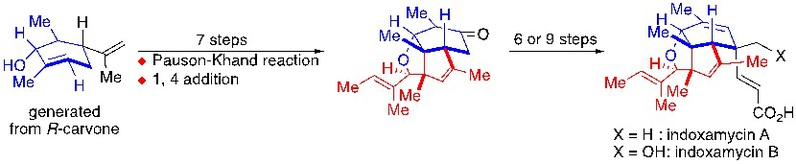
5) Hu, N.; Dong, C.; Zhang, C.; Liang, G.* “Total Synthesis of (–)-Indoxamycins A and B” Angew. Chem. Int. Ed. 2019,58, 6659.
6) Zhang, X.; Zhang, C.; Wang, X.; Liang, G.* “Mild Friedel–Crafts Reactions Enable a Robust Synthesis of Roseophilin” Org. Lett.2019,21, 3357.
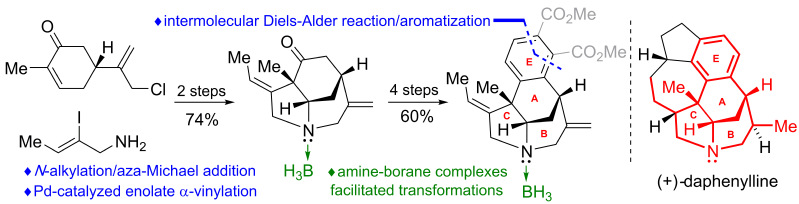
7) Deng, M.; Yao, Y.; Li, X.; Li, N.; Zhang, X.; Liang, G.* “Rapid Construction of the ABCE Tetracyclic Tertiary Amine Skeleton in Daphenylline Enabled by an Amine–Borane Complexation Strategy” Org. Lett.2019,21, 3290.
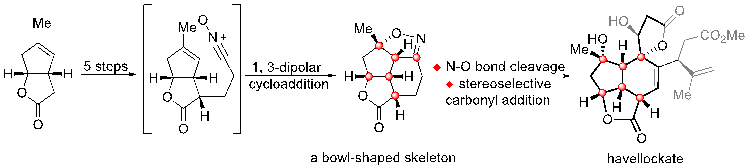
8) Deng, M.; Zhang, X.; Li, Z.; Chen, H.; Zang, S.; Liang, G.* “Rapid Construction of the Common [5–5–6] Tricyclic Ring Skeleton in Polycyclic Cembranoids and Nor-cembranoids via Intramolecular 1,3-Dipolar Cycloaddition” Org. Lett.2019,21, 1493.
9) Bi, X.; Xu, W.; Yao, Y.; Zhou, Li.; Liang, G.* “Total Syntheses of a Family of Cadinane Sesquiterpenes” J. Org. Chem. 2018, 83, 5825.
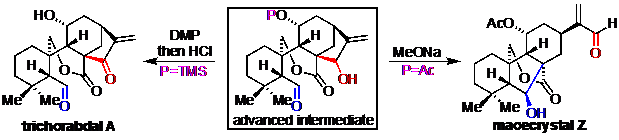
10) Lv, Z.; Chen, B.; Zhang, C.; Liang, G.* “Total Syntheses of Trichorabdal A and Maoecrystal Z” Chem. Eur. J. 2018, 24, 9773.
11) Yao, Y.; Wang, X.; Liang, G.* “Total Syntheses of (+)-Agelastatin A and (+)-Agelastatin B through Cationic Cyclizations”Tetrahedron2017, 73, 4538.
12) Geng, Q.; Xiao, X.; He, G.-R.; Yao, S.-M. Liang, G.* “One Stone Two Birds: Degradation of Persistent Organic Pollutants to a Valuable Industrial Chemical Production of Pentafluoropropionyl Fluoride from HFPO Oligomers Catalyzed by Cesium Fluoride in Tetraglyme” Chin. Chem. Lett.2016, 27, 1009.
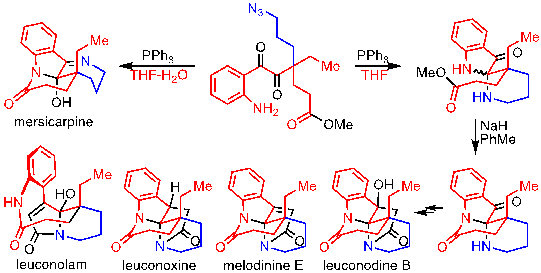
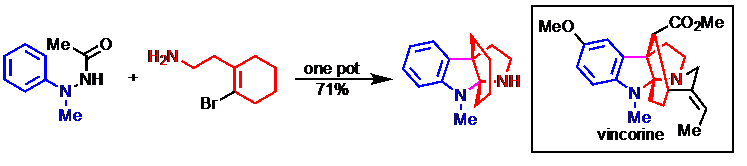
13) Geng, Q.; Li, Z.; Lv, Z.; Liang, G.* “Progress in Total Syntheses of Leuconolam-Leuconoxine-Mersicarpine Alkaloids” Chin. J. Org. Chem.2016, 36, 1447.
14) Qiao, T.; Liang, G.* “Recent Advances in Terpenoid Syntheses from China” Sci. Chin. Chem. 2016, 59, 1142.
15) Li, Z.; Geng, Q.; Lv, Z.; Pritchett, B. P.; Baba, K.; Numajiri, Y.; Stoltz, B. M.*; Liang, G.* “Selective Syntheses of Leuconolam, Leuconoxine, and Mersicarpine Alkaloids from a Common Intermediate through Regiocontrolled Cyclizations by Staudinger Reactions”Org. Chem. Front. 2015, 2, 236.
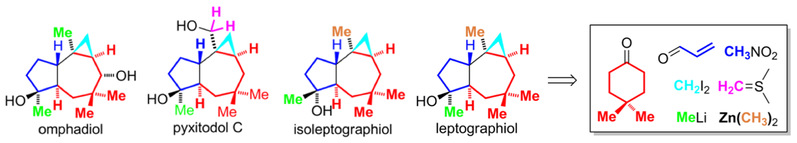
16) Zhou, L.; Yao, Y.; Xu, W.; Liang, G.* “Total Syntheses of (±)-Omphadiol and (±)-Pyxidatol C through a Cis-Fused 5,7-Carbocyclic Common Intermediate”J. Org. Chem. 2014,79, 5345.
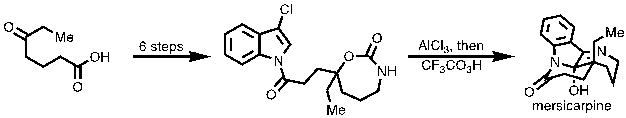
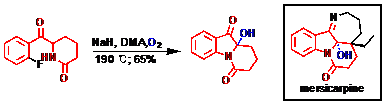
17) Lv, Z.; Li, Z.; Liang, G.* “Total Synthesis of Mersicarpine through a Cationic Cyclization Approach”Org. Lett.2014,16, 1653.
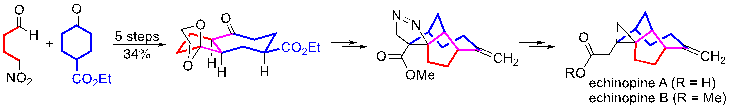
18) Xu, W.; Wu, S.; Zhou, L.; Liang, G.* “Total Syntheses of Echinopines”Org. Lett.2013, 15, 1978.
19) Li, Z.; Liang, G.* “Quick Access to the Core of Mersicarpine through an SNAr Strategy” Tetrahedron Lett. 2013, 54, 242.
20) Zhan, F.; Liang, G.* “Formation of Enehydrazine Intermediates through Coupling of Phenylhydrazines with Vinyl Halides: Entry into the Fischer Indole Synthesis”Angew. Chem. Int. Ed. 2013,52, 1266.
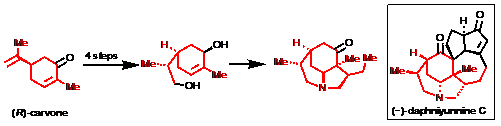
21) Yao, Y.; Liang, G.* “Rapid Construction of the ABC Ring System in the Daphniphyllum Alkaloid Daphniyunnine C”Org. Lett.2012,14, 5499.
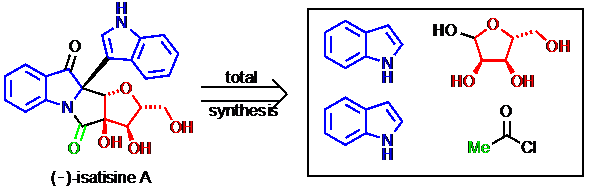
22) Zhang, X.; Mu, T.; Zhan, F.; Ma, L.; Liang, G.* “Total Synthesis of (–)-Isatisine A”Angew. Chem. Int. Ed.2011,50, 6164.
|
| 本组成员 |

敖军礼
外访生:南开大学博士研究生
邮箱:aoj190@163.com

臧绍丽
外访生:南开大学硕士研究生
邮箱:shaolizang@163.com

刘潇蓬
外访生:南开大学本科生
邮箱:1713636@mail.nankai.edu.cn

何鑫儿
暑期实习生/英国伯明翰大学本科毕业,秋季入学牛津大学研究生院
邮箱:XXH813@student.bham.ac.uk